24. Assertion :In B2H6, the terminal B H bonds are shorter, than
By A Mystery Man Writer
Description
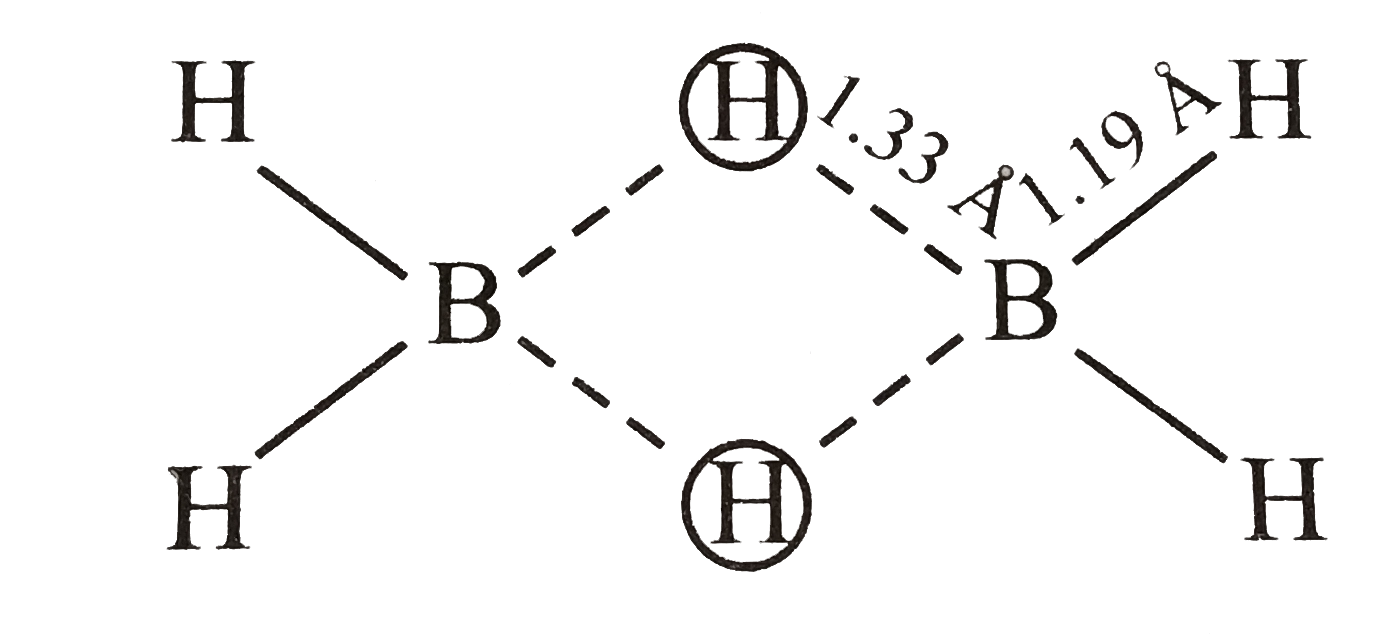
24. Assertion :In B2H6, the terminal B H bonds are shorter, than the B H bridge bonds Reason: The terminal B H bond order is greater than that of the B H bridge bond
24- Assertion-In B2H6- the terminal B-H bonds are shorter- than the B-H bridge bonds Reason- The terminal B-H bond order is greater than that of the B-H bridge bond
24- Assertion-In B2H6- the terminal B-H bonds are shorter- than the B-H bridge bonds Reason- The terminal B-H bond order is greater than that of the B-H bridge bond
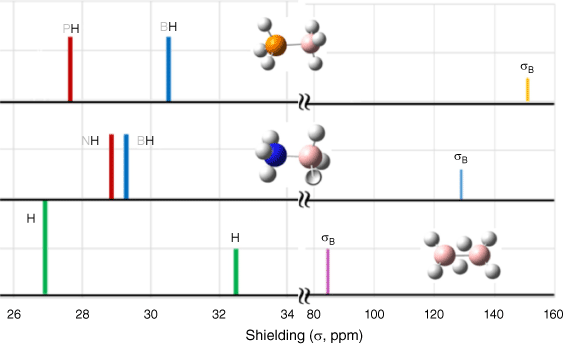
CSIRO PUBLISHING Australian Journal of Chemistry
Consider the following statements, (i) Diborane contains two centre

Is B-H-B banana bond in B2H6stronger than terminal B-H bond - Chemistry - The p-Block Elements - 6962819

The Source Function Descriptor as a Tool to Extract Chemical Information from Theoretical and Experimental Electron Densities
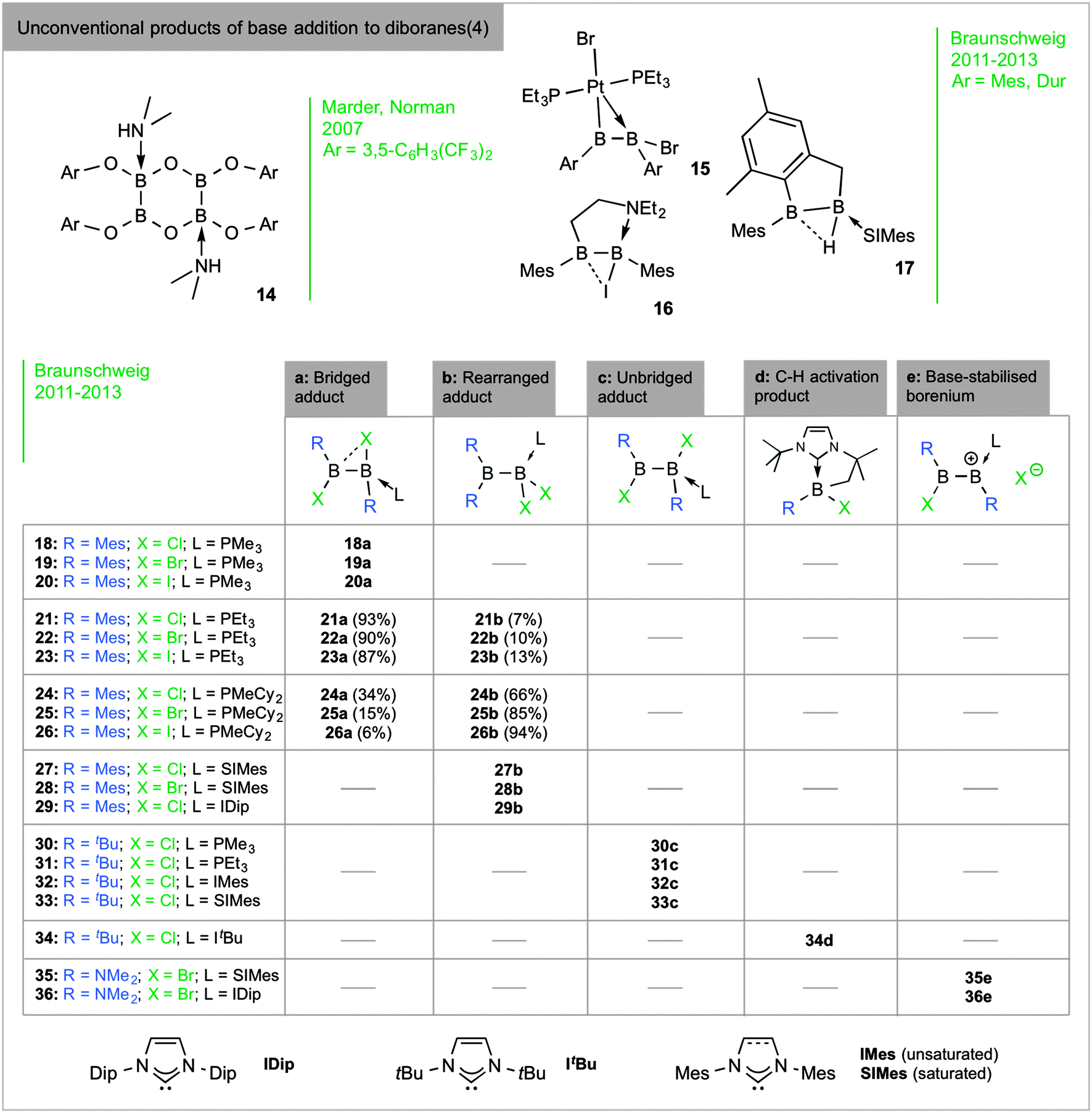
sp 2 –sp 3 diboranes: astounding structural variability and mild sources of nucleophilic boron for organic synthesis - Chemical Communications (RSC Publishing) DOI:10.1039/C5CC02316E

Formation and Reactivity of Electron‐Precise B−B Single and Multiple Bonds - Arrowsmith - 2017 - Angewandte Chemie International Edition - Wiley Online Library

Chemical Bonding SPECIAL ASSIGNMENT, PDF, Chemical Polarity

In which of the following compound (s) terminal (2C - 2e^{-}) bond and bridge bonds are lying in same plane

Why is bridge bond stronger but longer in diborane? - Quora
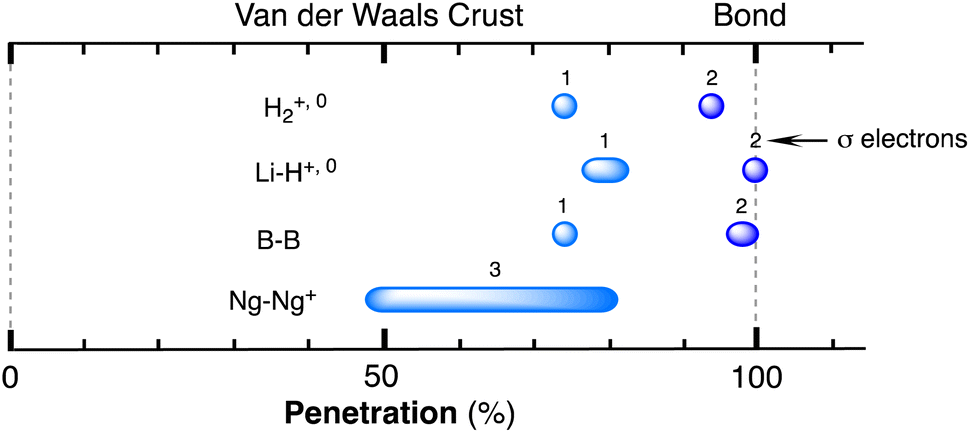
The borderless world of chemical bonding across the van der Waals crust and the valence region - Chemical Science (RSC Publishing) DOI:10.1039/D3SC02238B

ReasonHigher the bond order,shorter is the bond length and greater is the bond energy.AssertionBond order can assume any value including zero

The correct statement(s) regarding diborane (B_2H_6) is/are : (a) Maximum six hydrogen atoms can

Group 13 Flashcards
from
per adult (price varies by group size)