Solved 1. A cup of hot 50°C water is poured onto 35g of ice
By A Mystery Man Writer
Description
Answer to Solved 1. A cup of hot 50°C water is poured onto 35g of ice
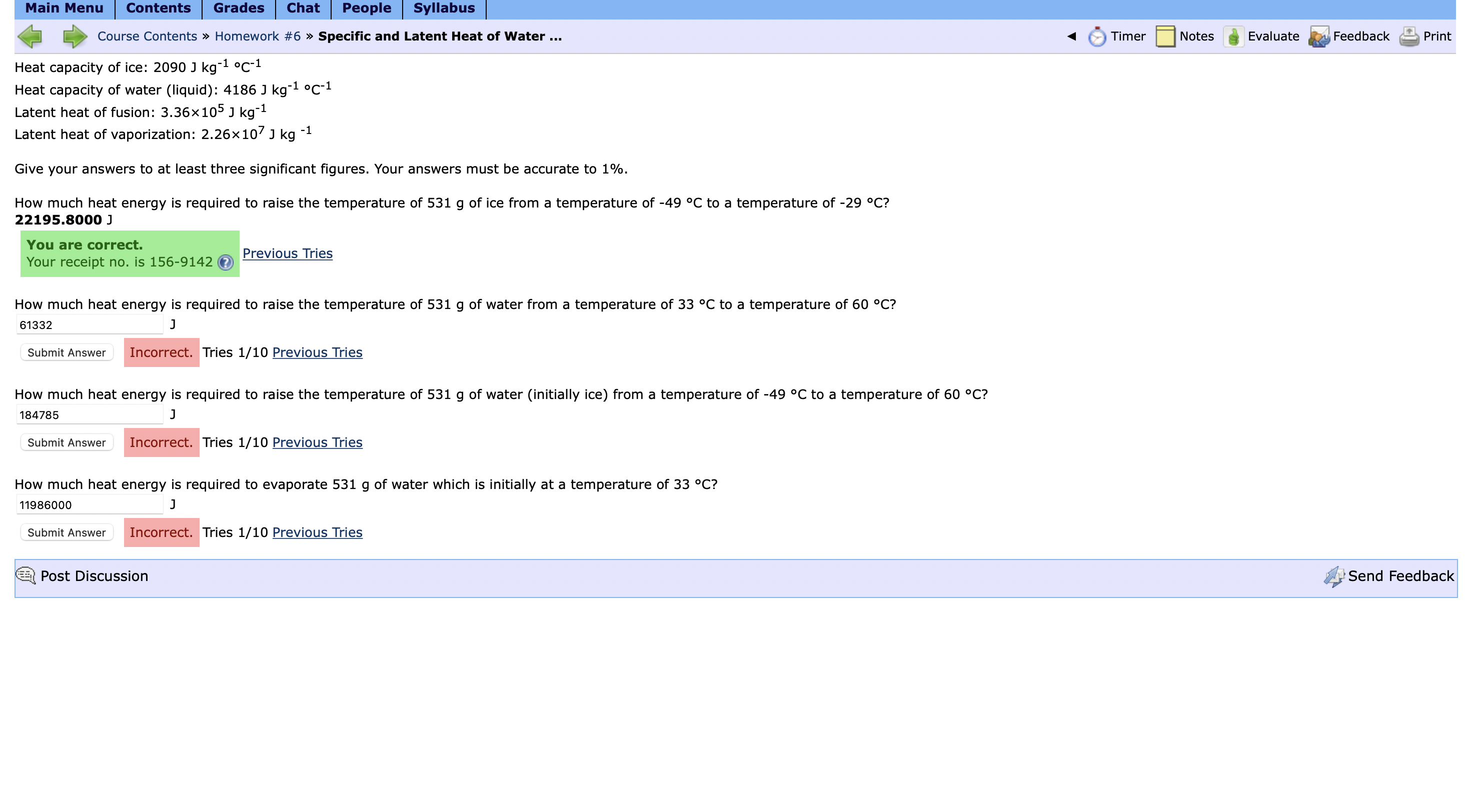
Solved Heat capacity of ice: 2090Jkg-1°C-1Heat capacity of
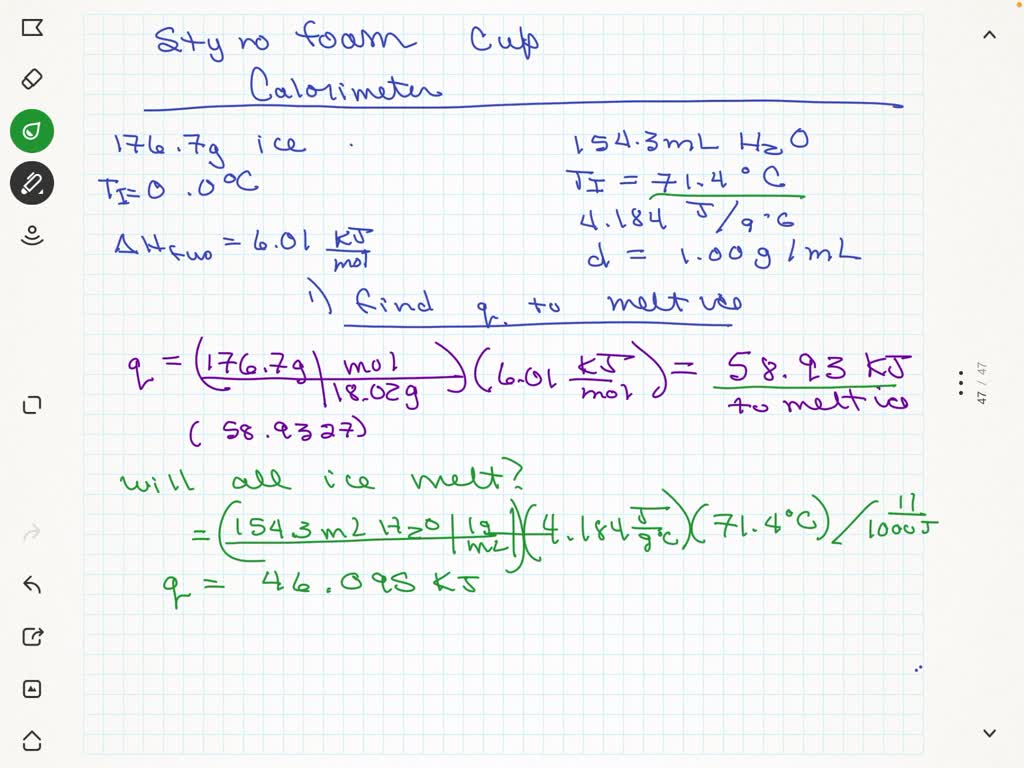
SOLVED: A 176.7 g sample of ice at 0.0 °C is added to 154.3 mL of liquid water at 71.4 °C in a styrofoam cup calorimeter. (The specific heat capacity of water

Solved - A 153.7 g sample of ice at 0.0 °C is added to 150.7

8.2: Calorimetry (Problems) - Chemistry LibreTexts
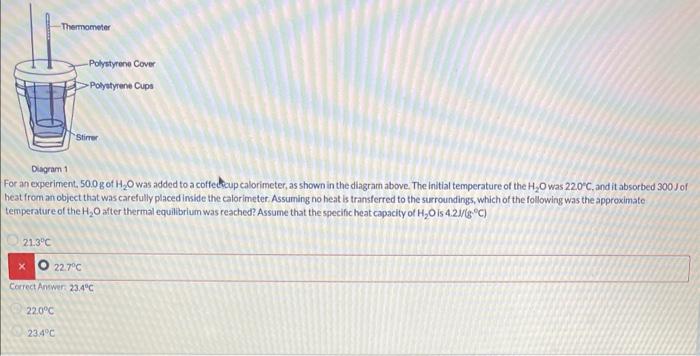
Solved For an experiment, 50.0 g of H2O was added to a

single serve dessert with 19 grams of quality protein 👇👇 🍫I put my

DIRTY WATER OF THE DAY: Lemon Meringue Pie #drink #water #watertok #wa, Lemon Meringue Pie

Sicilian gelato-style ice cream – ICE CREAM NATION

0 Calorie Italian Ice The flavor is entirely up to you! #ninjacreami #, ninja creamy recipes

SOLVED: A cup of hot 50°C water is poured onto 35g of ice at 0°C until the entire system ends up at 20°C. How much energy is needed to melt the 35g

guided practice - ABS Community Portal
A student weighs a piece of ice and then allows it to melt. In your opinion, what will be the weight of water and why? - Quora
from
per adult (price varies by group size)







