My publications - CHM 201-LECTURE IV-REAL GASES - Page 8 - Created with Publitas.com
By A Mystery Man Writer
Description
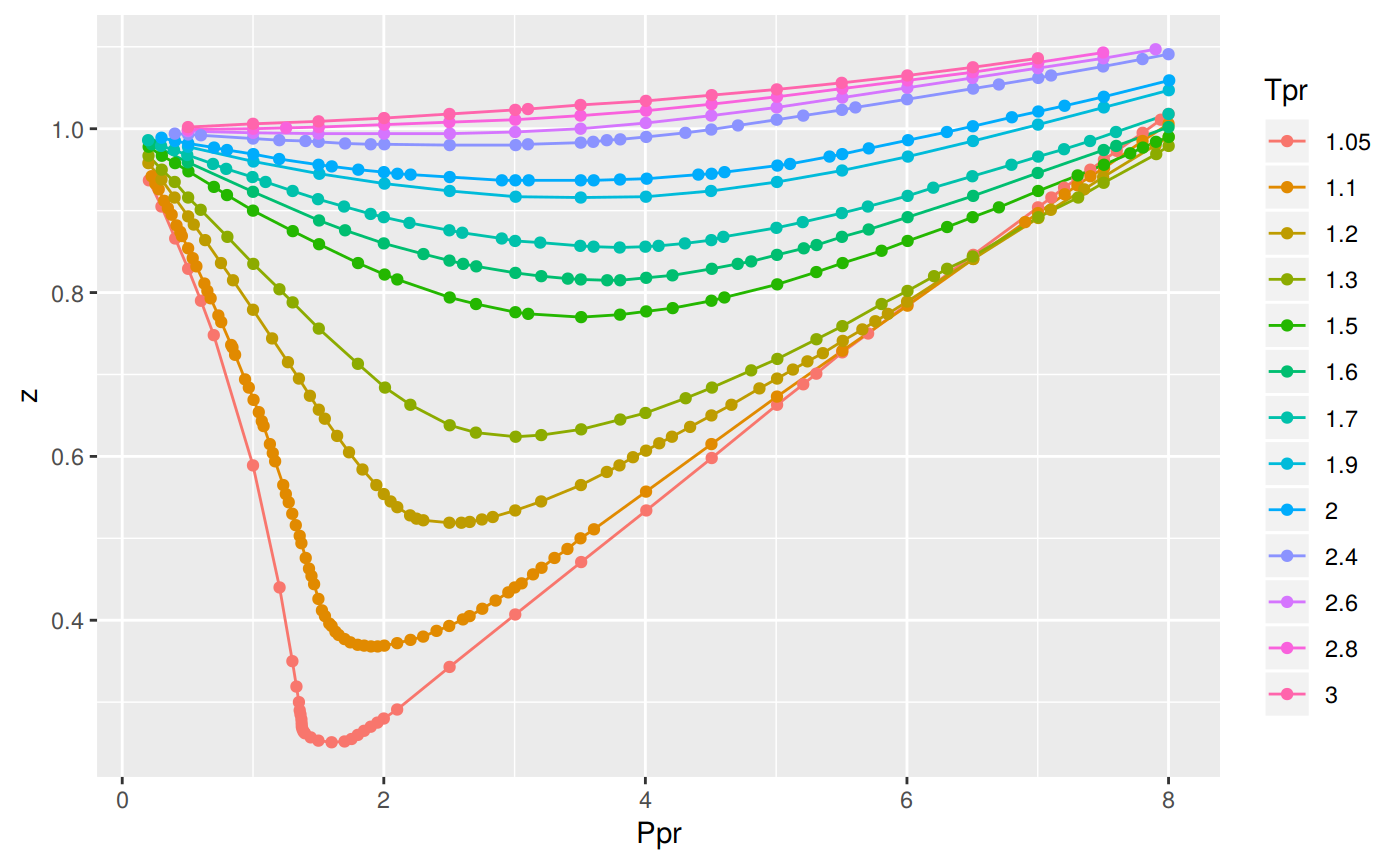
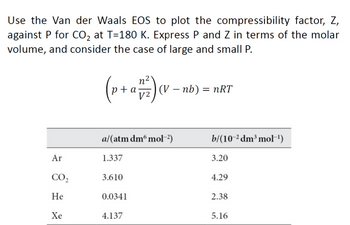
• Compressibility factor z: ▪ Dimensionless quantity. ▪ For a pure substance, it is a function of temperature and pressure or temperature and molar volume. ▪

SOLUTION: Gaseous state practice sheet gaseous state chemistry

PDF) Identifying and Engaging the Internal and External Stakeholders, the Outcome and Target Champions and Collaborators

My publications - CHM 201-LECTURE IV-REAL GASES - Page 8 - Created
Chem 201: General Chemistry I OER - Chemistry LibreTexts

Free Chemistry Textbook Available for Download - OpenStax

1.2.6 Real Gases, DP IB Chemistry: SL Revision Notes 2016

CHEM 201] - Final Exam Guide - Ultimate 59 pages long Study Guide
Chemistry: A Study of Matter

My publications - CHM 201-LECTURE IV-REAL GASES - Page 27

PDF) Identifying and Engaging the Internal and External Stakeholders, the Outcome and Target Champions and Collaborators

General Chemistry 1B. Lecture 4. Intermolecular Forces Liquids & Solids, Part IV

Abhijit Roy - Performing Democracy in Culture and Politics in South Asia - Performative Communication-Routledge (2018)
from
per adult (price varies by group size)