Developing a Thermodynamical Method for Prediction of Activity Coefficient of TBP Dissolved in Kerosene
By A Mystery Man Writer
Description
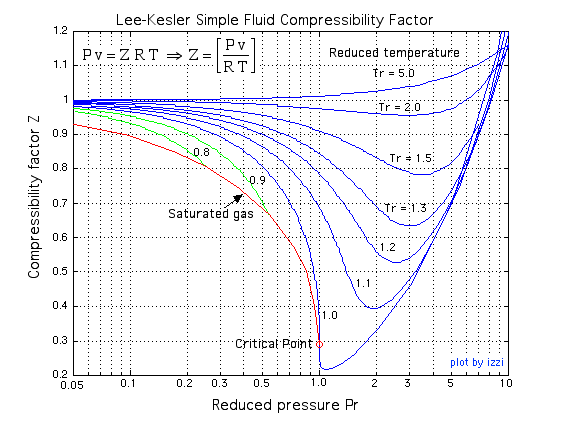
Results of the experimental measurements on the partial molar volume of kerosene used as a medium for dissolving TBP are utilized to determine the activity of TBP in the binary kerosene-TBP solution through the application of Gibbs-Duhem equation. The treatment is based on combination of the experimental data with the thermodynamic values available on the compressibility factor of pure kerosene at room temperature. It is shown that the activity of TBP in kerosene has a positive deviation from ideality with an activity coefficient derived as follows:1) at X TBP ≤ 0.01: γ TBP = 42.530, 2) at the 0.01 X TBP 0.2: 3) at the higher TBP concentrations 0.2 X TBP 0.97: and 4) at TBP Raoultian concentrations 0.97 ≤ X TBP:γ TBP = 1. These quantities can be utilized at temperature closed to 298 K.

Water adsorption in the organic phase for the D2EHPA-kerosene

Liquid–Liquid Equilibrium Data of Extraction with Tri-n-butyl Phosphate on Phosphoric Acid Containing Sodium Sulfate or Potassium Sulfate
Sustainability, Free Full-Text
All abstracts

A comparison between TEHA and Cyanex 923 on the separation and the

Eskandar Keshavarz Alamdari - PDF Free Download

Full article: Nitric Acid Extraction into the TODGA/TBP Solvent

Developing a Thermodynamical Method for Prediction of Activity

PDF] EXTRACTION OF ZN, MN AND CO FROM ZN-MN-CO-CD-NI CONTAINING

E. ALAMDARI, Professor (Associate), PhD

Vidal. Thermo. App. in Chem. Eng, PDF, Enthalpy

The experimental investigations on viscosity, surface tension, interfacial tension and solubility of the binary and ternary systems for tributyl phosphate (TBP) extractant in various organic solvents with water: Thermodynamic NRTL model and

PDF) Developing a Thermodynamical Method for Prediction of

All abstracts
from
per adult (price varies by group size)