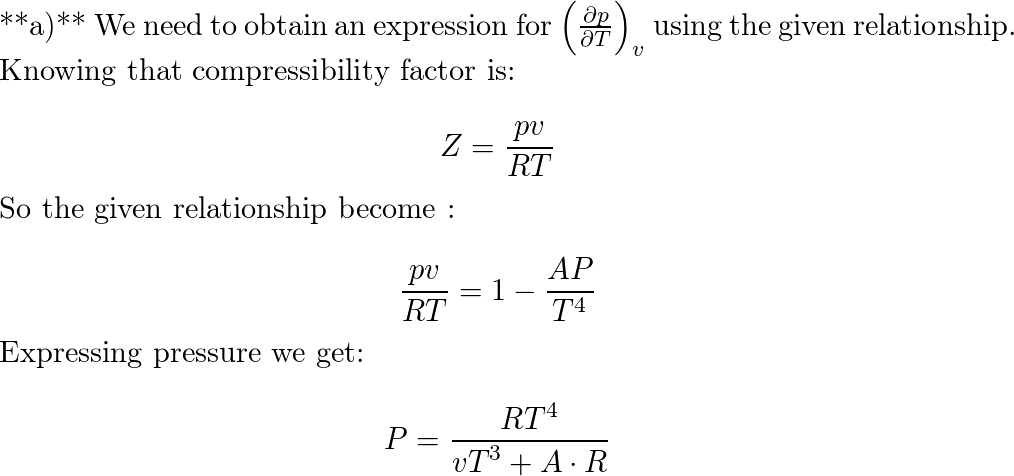Developing a Thermodynamical Method for Prediction of Activity
By A Mystery Man Writer
Description
Results of the experimental measurements on the partial molar volume of kerosene used as a medium for dissolving TBP are utilized to determine the activity of TBP in the binary kerosene-TBP solution through the application of Gibbs-Duhem equation. The treatment is based on combination of the experimental data with the thermodynamic values available on the compressibility factor of pure kerosene at room temperature. It is shown that the activity of TBP in kerosene has a positive deviation from ideality with an activity coefficient derived as follows:1) at X TBP ≤ 0.01: γ TBP = 42.530, 2) at the 0.01 X TBP 0.2: 3) at the higher TBP concentrations 0.2 X TBP 0.97: and 4) at TBP Raoultian concentrations 0.97 ≤ X TBP:γ TBP = 1. These quantities can be utilized at temperature closed to 298 K.

Separation of Re and Mo from Roasting-Dust Leach-Liquor Using Solvent Extraction Technique by TBP

Molecular thermodynamic model for solvent extraction of mineral acids by tri-n-butyl phosphate (TBP) - ScienceDirect

DeepTM: A deep learning algorithm for prediction of melting temperature of thermophilic proteins directly from sequences - Computational and Structural Biotechnology Journal

Water adsorption in the organic phase for the D2EHPA-kerosene/water and aqueous Zn2+, CO2+, Ni2+ sulphate systems

Synergistic effect of MEHPA on co-extraction of zinc and cadmium with DEHPA

PDF) Machine Learning in Thermodynamics: Prediction of Activity Coefficients by Matrix Completion
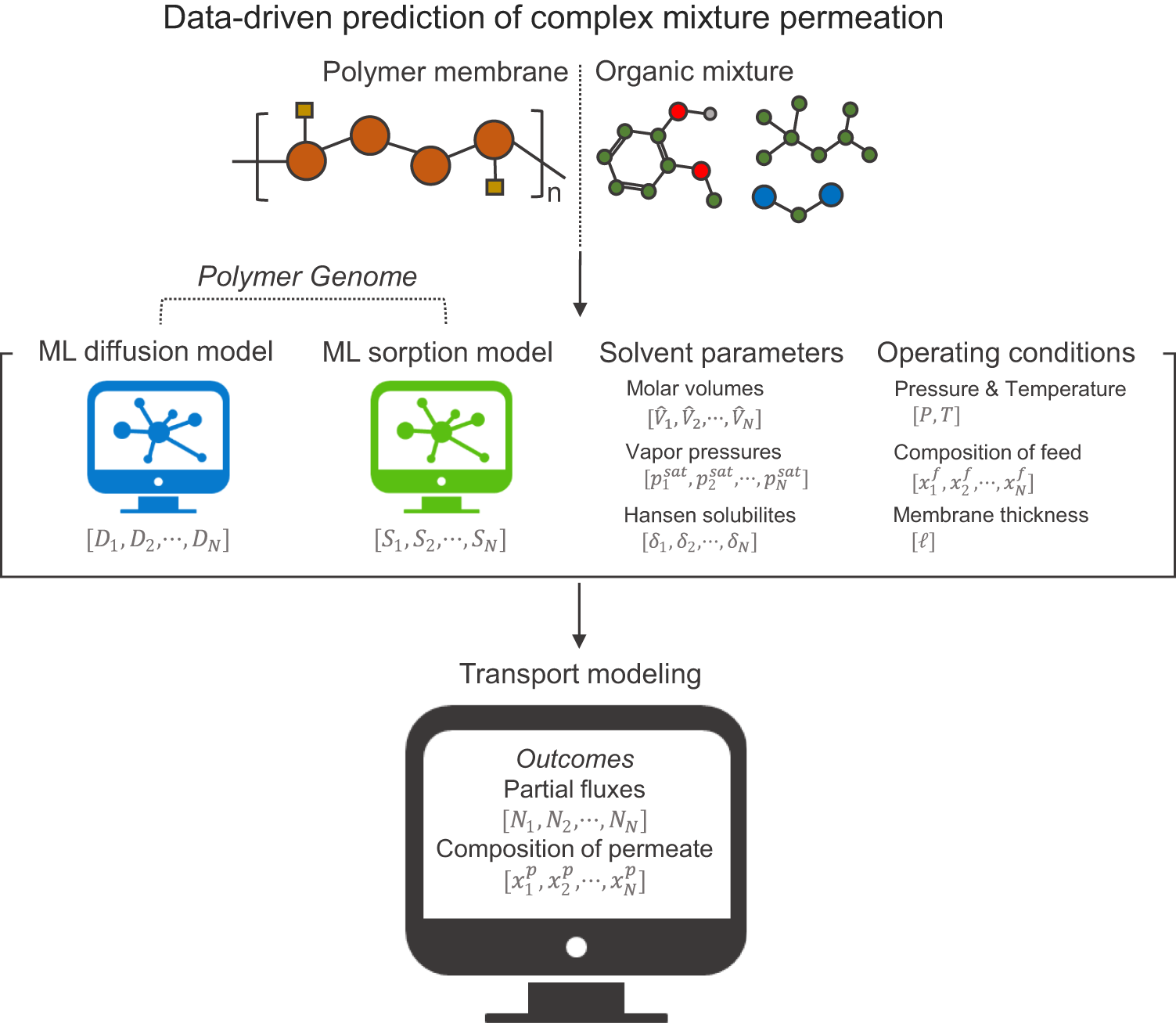
Data-driven predictions of complex organic mixture permeation in polymer membranes
New thermodynamic activity-based approach allows predicting the feasibility of glycolysis

D2EHPA with Cyanex 272 and individual D2EHPA. Overall flow sheet for

Thermodynamics and Property Models for Chemical Systems

PDF) Developing a Thermodynamical Method for Prediction of Activity Coefficient of TBP Dissolved in Kerosene
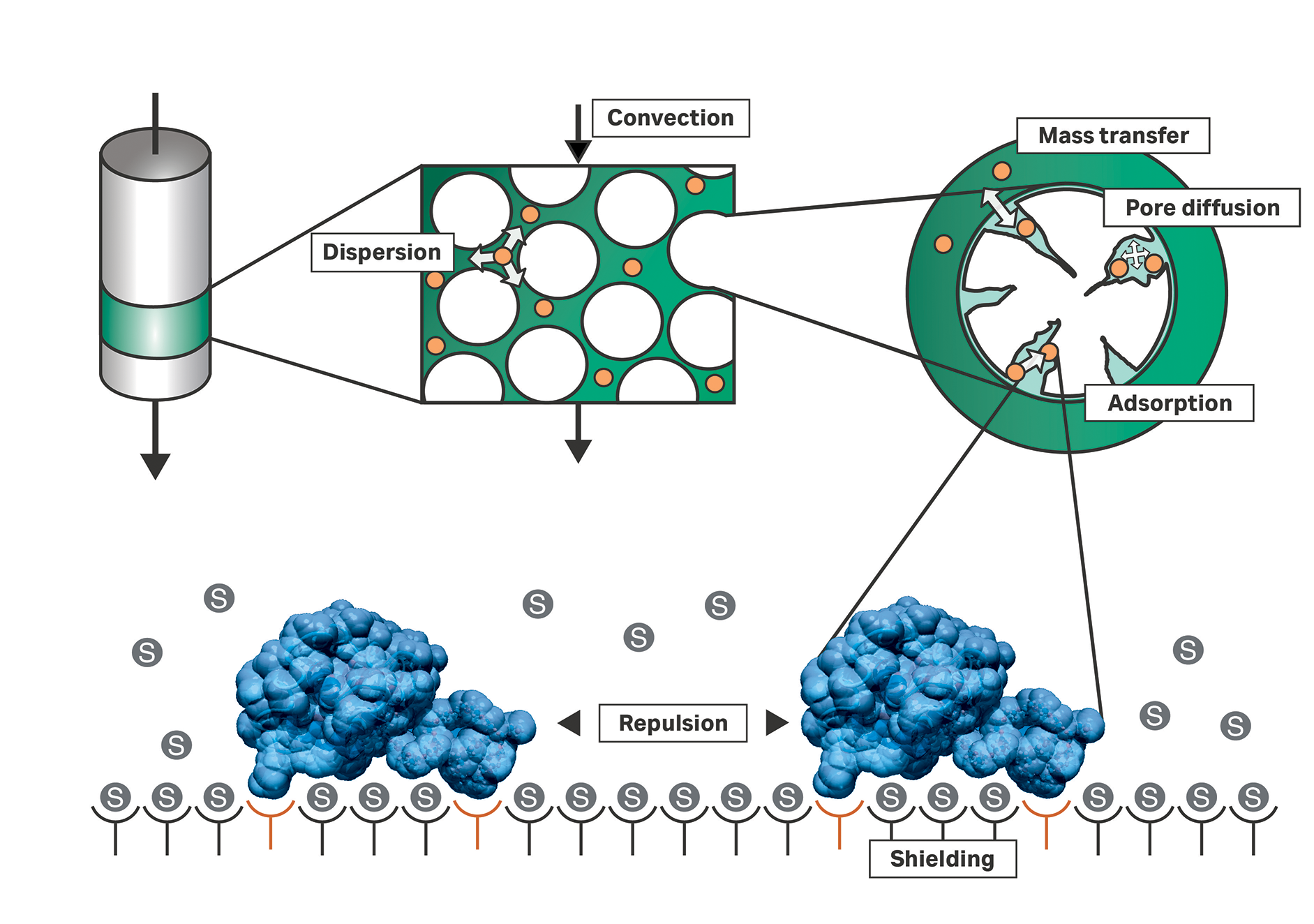
Mechanistic modeling-Digital twins are here to stay

PDF) Developing a Thermodynamical Method for Prediction of Activity Coefficient of TBP Dissolved in Kerosene

PDF) The application of thermodynamic methods in drug design

The integral molar volume of TBP-kerosene binary solution as a function
from
per adult (price varies by group size)