20.If Z is a compressibility factor, van der Waals equation at low pressure can be written as
By A Mystery Man Writer
Description
20.If Z is a compressibility factor, van der Waals equation at low pressure can be written as
20-If Z is a compressibility factor- van der Waals equation at low pressure can be written as
20-If Z is a compressibility factor- van der Waals equation at low pressure can be written as

physical chemistry - Why do some gases have lower value of Z for a particular pressure? - Chemistry Stack Exchange
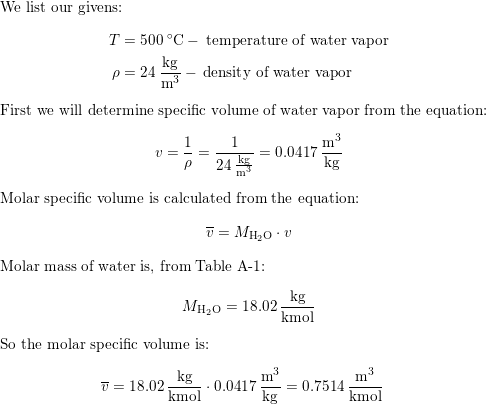
Estimate the pressure of water vapor at a temperature of $50
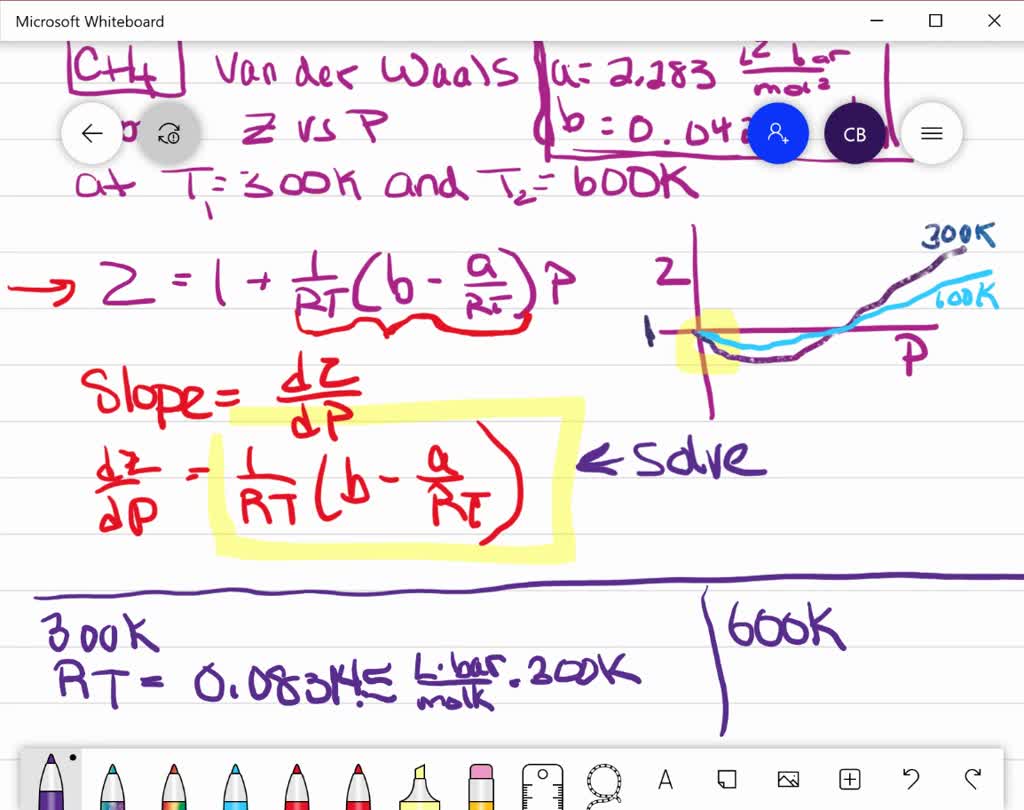
⏩SOLVED:Use the van der Waals constants for CH4 in Table 1.3 to…

At low pressure the van der Waals' equation is reduced to [P +(a)/(V^(
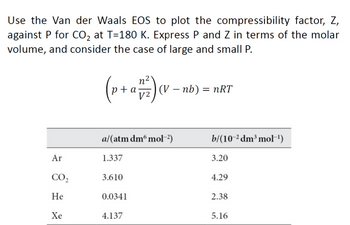
Answered: Use the Van der Waals EOS to plot the…

At low pressure, the compressibility factor is given as

The compressiblity factor a gas obeying van der Waals' equation of state is given by V V-b RTV (2) a ✓ RTV V-b V-b RTV (3) Va (4) RTV V-6

Van der Waals Equation - Derivation, Relation Between Ideal Gas Law, Application

Solved The Van Der Waals equation of state is given by
from
per adult (price varies by group size)







