200 g of a sample of limestone liberates 66 g of CO2 on heating. The percentage purity of CaCO3 in the limestone is Options:a 95
By A Mystery Man Writer
Description
200 g of a sample of limestone liberates 66 g of CO2 on heating. The percentage purity of CaCO3 in the limestone is Options:a 95
200 g of a sample of limestone liberates 66 g of CO2 on heating- The percentage purity of CaCO3 in the limestone is Options-a- 95
200 g of a sample of limestone liberates 66 g of CO2 on heating- The percentage purity of CaCO3 in the limestone is Options-a- 95

kemy11a by arta asad - Issuu

6.5 g of an impure sample of limestone liberates 2.2 g of CO2 on strong heating. The percentage purity of
200g of a sample of limestone liberates 66 g of co2 on heating. The

SOLVED: A sample of limestone and other soil materials was heated, and the limestone decomposed to give calcium oxide and carbon dioxide. CaCO3 (s) â†' 3 CaO(s) + CO2 (g) A 6.631
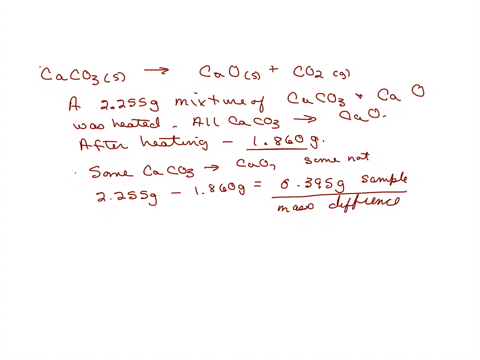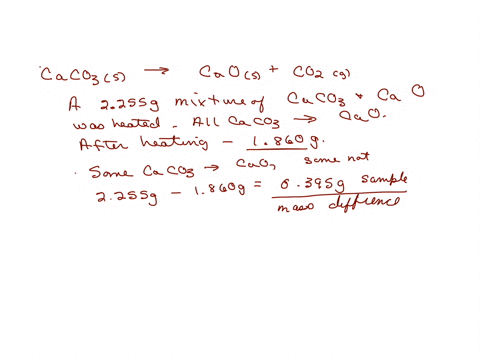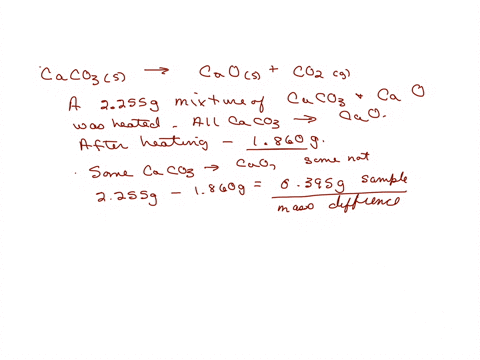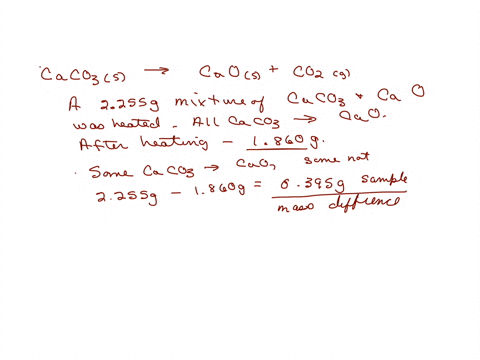
SOLVED: The weight percentage of limestone is as follows: CaCO3 = 93%, MgCO3 = 5%, Insoluble = 2%. This limestone is burned in a vertical kiln to produce lime (CaO, MgO, Insolubles).

Solved A limestone analysis: CaCO, 92.89% MgCO3 5.41%

PDF) Valorization of waste powders from cement-concrete life cycle: A pathway to circular future
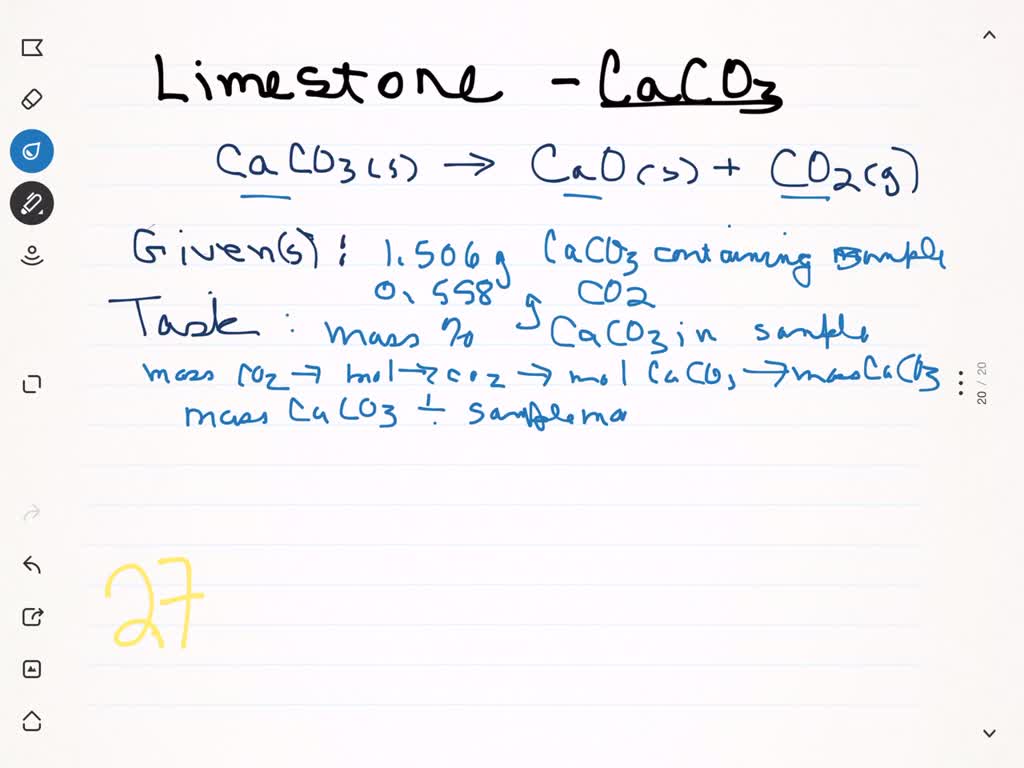
⏩SOLVED:A sample of limestone and other soil materials was heated,…
Integrated CO 2 capture and electrochemical upgradation: the underpinning mechanism and techno-chemical analysis - Chemical Society Reviews (RSC Publishing) DOI:10.1039/D2CS00512C

Solved Please help me solve the following questions below

Energies, Free Full-Text
from
per adult (price varies by group size)