Solved An ideal gas initially at Pi, Vi, and Ti is taken
By A Mystery Man Writer
Description

First Alert Radon Gas Test Kit, RD1

SOLVED: An ideal gas initially at Pi, Vi, and Ti is taken through a cycle as shown below. (Let the factor n = 3.1.) (a) Find the net work done on the

⏩SOLVED:An ideal gas initially at Pi, Vi, and Ti is taken through a…
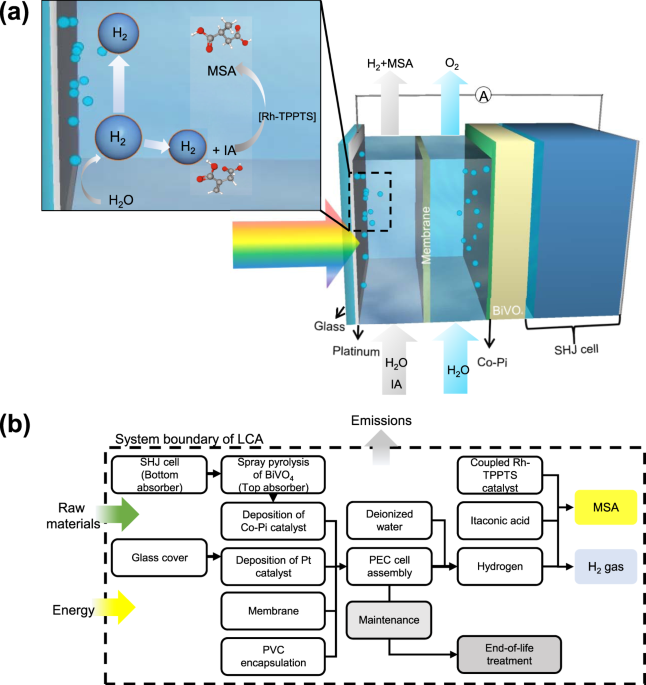
Life cycle net energy assessment of sustainable H2 production and hydrogenation of chemicals in a coupled photoelectrochemical device

SOLVED: An ideal gas initially at Pi, Vi, and Ti is taken through a cycle as shown in the figure (Pressure in Pa and Volume in m^3) What is the net energy
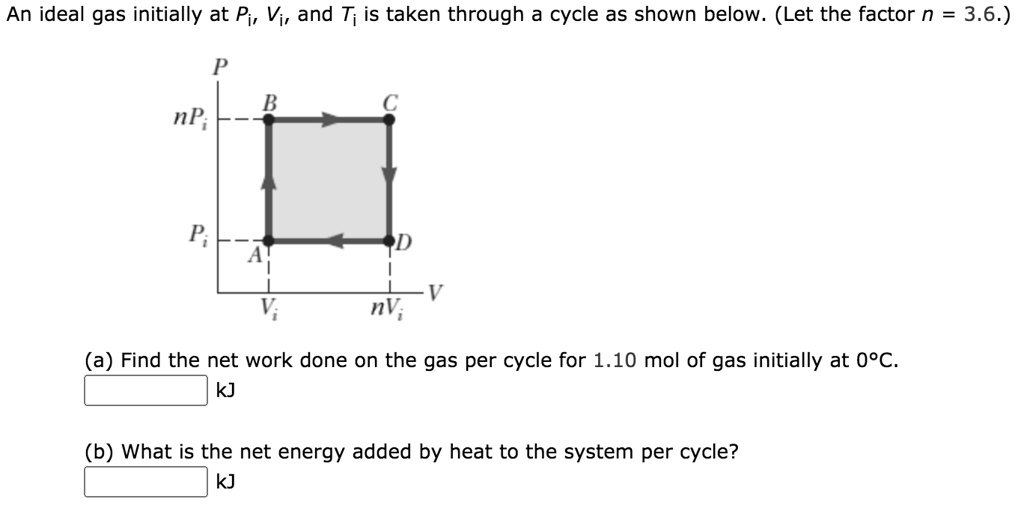
SOLVED: An ideal gas initially at Pi, Vi, and Ti is taken through a cycle as shown below. (Let the factor n 3.6.) nP; P; nV; (a Find the net work done
SOLVED: 3P; 3Vi A mole of ideal gas initially at Pi-l Pa, Vi-S m³, and Ti= 0°C is taken through a cycle as shown in the above Figure. a) Find the temperature

Relating Pressure, Volume, Amount, and Temperature: The Ideal Gas Law

An ideal gas initially P_i ,V_i , and T_i is taken through a cycle as shown in Figure. (a) Find the net work done on the gas per cycle 1.00 mol of

SOLVED: An ideal gas initially at Pi, Vi, and Ti is taken through a cycle as shown below. (Let the factor n 3.6.) nP; P; nV; (a Find the net work done

Solved 2. One mole of an ideal gas is initially at pressure
from
per adult (price varies by group size)