The entropy change for the conversion of 36 g water to vapour at
By A Mystery Man Writer
Description
The entropy change for the conversion of 36 g water to vapour at its boiling point at 1 atm is (Enthalpy of vaporization for water is 40.63 kJ mol–1)
The entropy change for the conversion of 36 g water to vapour at its boiling point at 1 atm is -Enthalpy of vaporization for water is 40-63 kJ mol-1
The entropy change for the conversion of 36 g water to vapour at its boiling point at 1 atm is -Enthalpy of vaporization for water is 40-63 kJ mol-1

JEE Main Previous Year Questions (2016- 2023): Thermodynamics - 35 Years Chapter wise Previous Year Solved Papers for JEE PDF Download
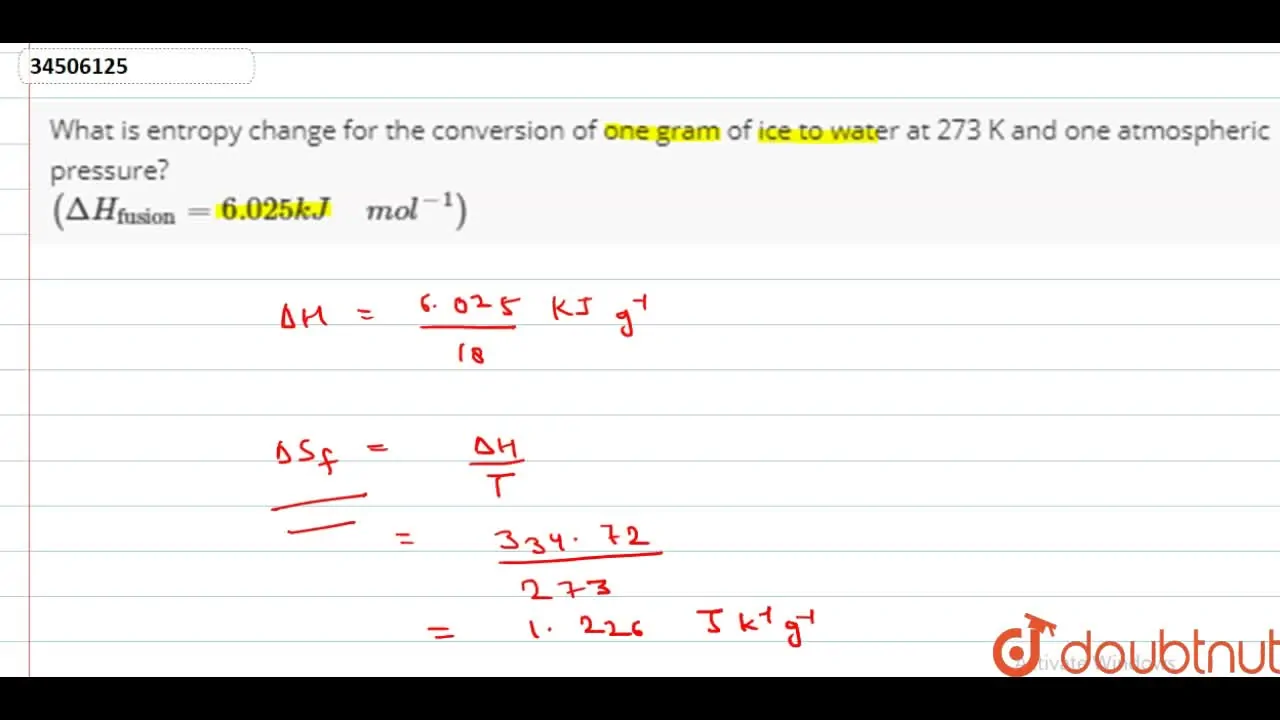
What is entropy change for the conversion of one gram of ice to water

Calculate the entropy change of n-hexane when 1 mole of it evaporates

Enthalpy of vaporization - Wikipedia
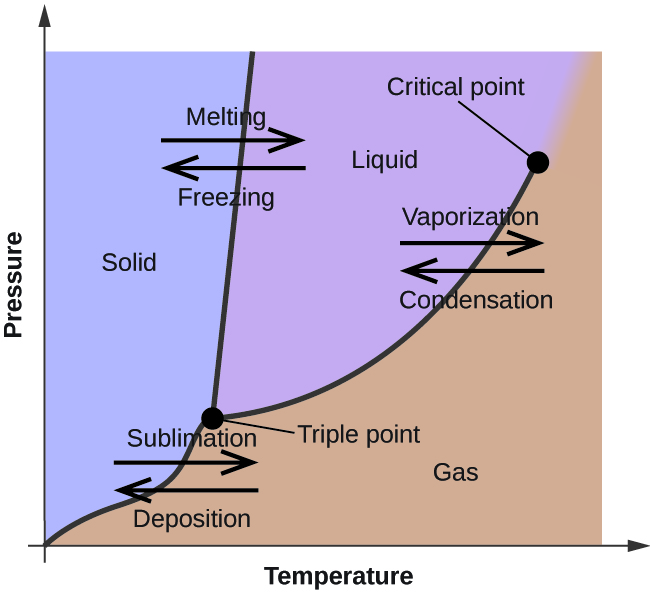
Phase Diagrams Chemistry for Majors
Why is the change in entropy of the vaporization of water 0 at 373 K? - Quora

66. The entropy change for the conversion of 36 g of water to vapour at 100°C (Normal boiling point) is

The entropy change when 36g of water evaporates at 373 K is :- (DeltaH

Calculate the entropy change involved in the vaporisation of water at

Phase transition - Wikipedia

The entropy change when 36g of water evaporates at 373 K is :- (DeltaH

50. Ir Water vapour is assumed to be a perfect gus, molar enthalpy change vapourisation of 1 mole of water 1 bar and 100° C is 41 mol Calculate the internal energy

The entropy change associated with the conversion of 1 kg of ice at 27
from
per adult (price varies by group size)






