At 273 K measurements on argon gave B = -21.7 cm$^3$ mol$^{
By A Mystery Man Writer
Description

Chap1

Solved 6. Take the van der Waals equation RT a 2 a. Express
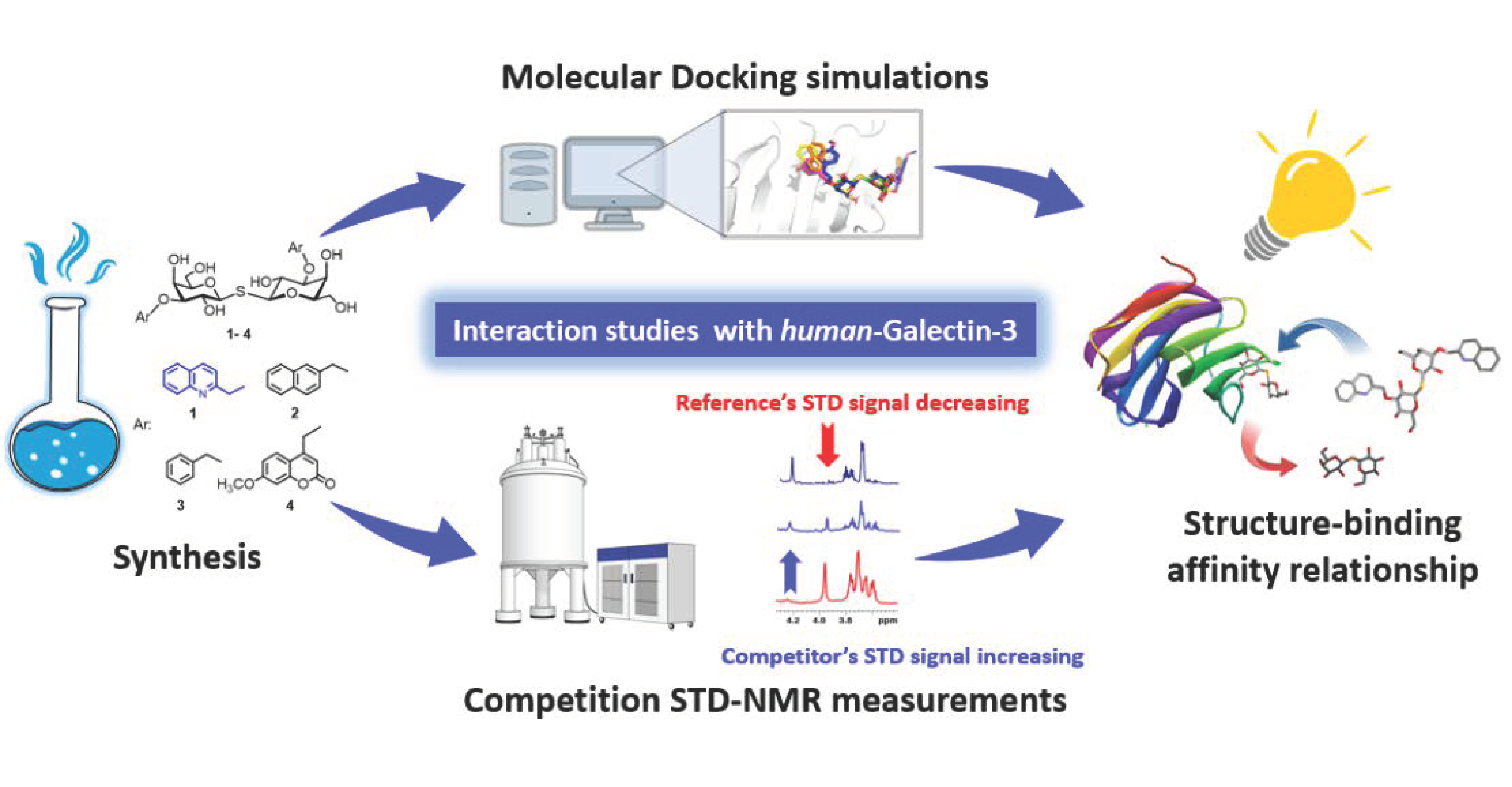
IJMS, Free Full-Text

Advanced carbon molecular sieve membranes derived from molecularly engineered cross-linkable copolyimide for gas separations

Answered: Set up the following conversion…

a) Third virial coefficient of argon. Experimental data: ▽, Michels et

At 273 K and 1 atm pressure, 1 mol of an ideal gas occupies 22.4

Molarity & Stoichiometry, Definition, Formula & Calculation - Lesson

Chemistry - Unit 3 - Joseph Flashcards

SOLVED: (9.5 Measurements of the viscosity; n, of argon gas 40 Ar) over a range of pressures yield the follow- ing results at two temperatures: at 500 K 3.5 X 10-5 kg

SOLVED: At 273 K, measurements on argon gave B = -21.7 cm^3/mol and C = 1200 cm^6/mol^2, where B and C are the second and third virial coefficients in the expression of

Consider the following samples of gases: If the three samples ar
from
per adult (price varies by group size)







