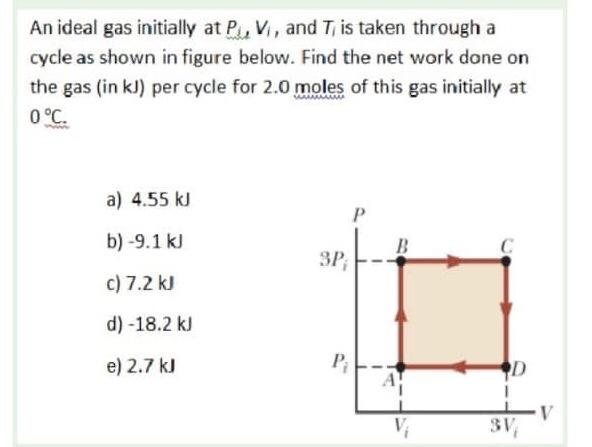
An ideal gas initially P_i ,V_i , and T_i is taken through a cycle as shown in Figure. (a) Find the net work done on the gas per cycle 1.00 mol of
By A Mystery Man Writer
Description
Click here:point_up_2:to get an answer to your question :writing_hand:an ideal gas initially at pi vi and ti is taken through a cycle
Click here👆to get an answer to your question ✍️ An ideal gas initially P-i -V-i - and T-i is taken through a cycle as shown in Figure- -a- Find the net work done on the gas per cycle 1-00 mol of gas initially 0-0C- -b- What is the net energy added by heat to the gas per cycle
Click here👆to get an answer to your question ✍️ An ideal gas initially P-i -V-i - and T-i is taken through a cycle as shown in Figure- -a- Find the net work done on the gas per cycle 1-00 mol of gas initially 0-0C- -b- What is the net energy added by heat to the gas per cycle

Chapter 19 The Second Law of Thermodynamics
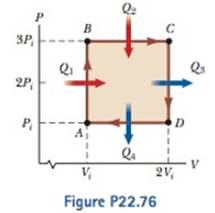
A 1.00-mol sample of a monatomic ideal gas is taken through the cycle shown in Figure P22.76. At point A, the pressure, volume, and temperature are P i , V i
Solved An ideal gas initially at PJ, V, and Ti is taken

11/14/2013PHY 113 C Fall Lecture 221 PHY 113 C General Physics I 11 AM – 12:15 PM MWF Olin 101 Plan for Lecture 22: Chapter 21: Ideal gas equations. - ppt download

Molecules August-1 2022 - Browse Articles

PDF) Advanced Thermodynamics for Engineers

SOLVED: An ideal gas initially at Pi' Vi' and Ti is taken through a cycle as shown below. (Let the factor n 2.8.) nP; nV; a) Find the net work done on

⏩SOLVED:An ideal gas initially at Pi, Vi, and Ti is taken through a…
Solving a Monatomic Ideal Gas Expansion Problem

amp; (%)

21 - The Kinetic Theory of Gases - Direito Processual Civil I

PDF) Chapter 18 Heat and the First Law of Thermodynamics Conceptual Problems
from
per adult (price varies by group size)







