117. Compressibility factor H, behaving as rea gas is 1) 1 RTV 3) 1+- RT 4) (1-a) 18. If V is the observed molor unlum
By A Mystery Man Writer
Description
Click here:point_up_2:to get an answer to your question :writing_hand:117 compressibility factor for h behaving as reagas is1 1rtv31rt41a18 if v is the observed
Click here👆to get an answer to your question ✍️ 117- Compressibility factor H- behaving as rea gas is 1- 1 RTV 3- 1- RT 4- -1-a- 18- If V is the observed molor unlum
Click here👆to get an answer to your question ✍️ 117- Compressibility factor H- behaving as rea gas is 1- 1 RTV 3- 1- RT 4- -1-a- 18- If V is the observed molor unlum

Magnesium based materials for hydrogen based energy storage: Past, present and future - ScienceDirect
IPCB Publications Database

117. Compressibility factor H, behaving as rea gas is 1) 1 RTV 3) 1+- RT 4) (1-a) 18. If V is the observed molor unlum

An unknown gas at 37.1 ∘c and 1.00 atm has a molar mass of 30.07 g/mol. assuming ideal behavior, what is

Non-Ideal Gas Behavior Chemistry: Atoms First

Solved The plot below shows how compressibility factor (Z)
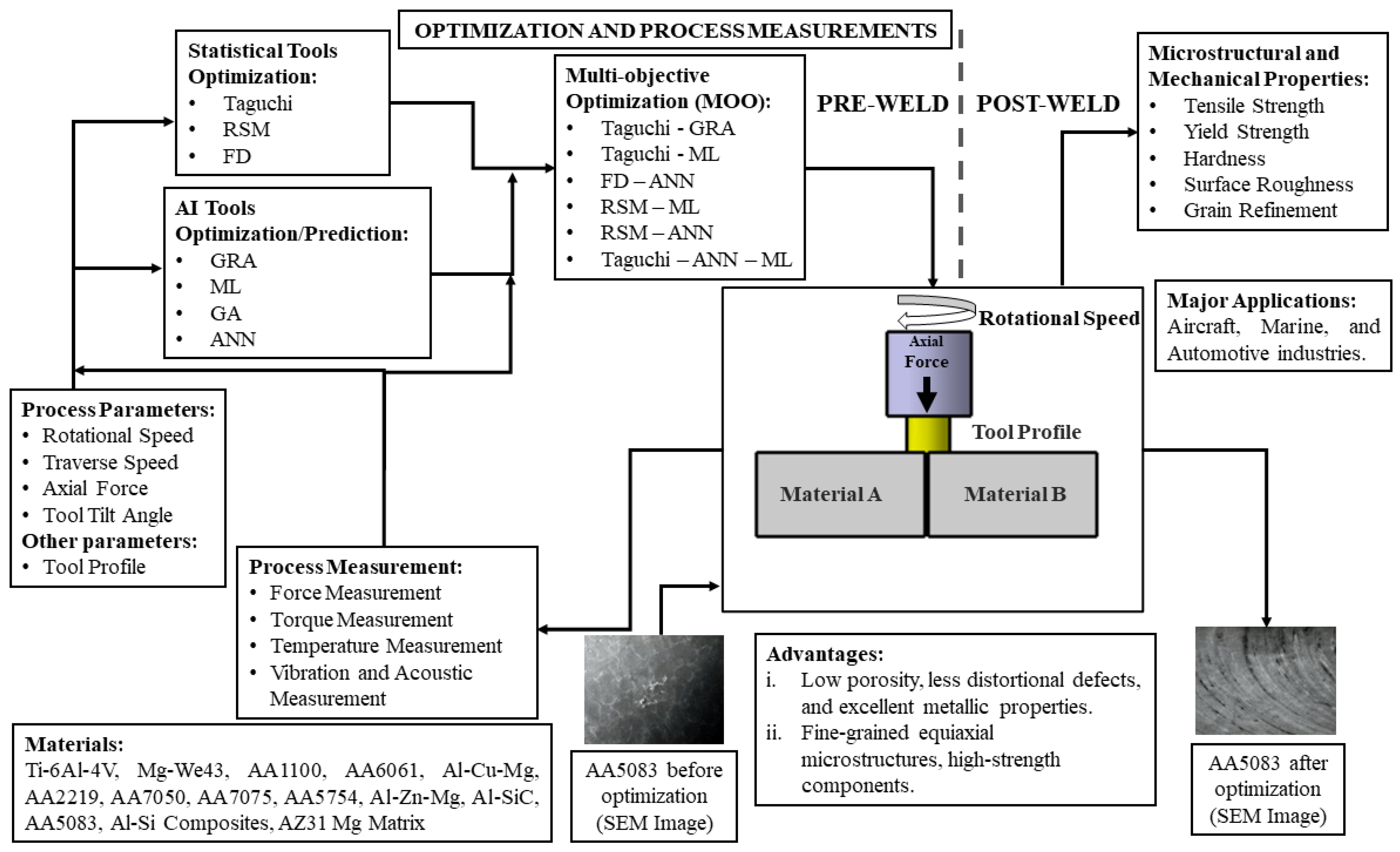
JMMP, Free Full-Text

At low pressure, the compressibility factor of one mole of gas can be written as Brand pramu mb) sono (1) 1-9 271 RTV (3) 1-RT (4) 1- av hinh of the following has maximum numbertof F

download - iccf

A comprehensive survey of research towards AI-enabled unmanned aerial systems in pre-, active-, and post-wildfire management

The compressibility factor a real gas is BP expressed by, Z=1+ er. The value of B 500 K and 600 bar is 0.0169 L/mol. Therefore the molar volume of the gas 500
from
per adult (price varies by group size)






