An ideal gas is taken from (Pi, Vi) to (Pf, Vf) in three different ways. Identify the process in which the work done on the gas the most. - Physics
By A Mystery Man Writer
Description
An ideal gas is taken from (Pi, Vi) to (Pf, Vf) in three different ways. Identify the process in which the work done on the gas the most.

P-V diagram of an ideal gas is as shown in the given figure. Work done by the gas in the process
An ideal gas is taken fromPiVitoPfVfin three different ways Identify the process in which the work done on the gas the most

The First Law of Thermodynamics and Some Simple Processes

The origin of irreversibility and thermalization in thermodynamic processes - ScienceDirect

to accompany Physics Including Human Applications Fuller

Thermodynamics and Statistical Mechanics - Physics - University of

PDF) PRACTICAL CONVERSION OF ZERO-POINT ENERGY: FEASIBILITY STUDY OF ZERO-POINT ENERGY EXTRACTION FROM THE QUANTUM VACUUM FOR THE PERFORMANCE OF USEFUL WORK

Physics Web Assign Ch 12 #8
Five moles of an ideal gas are compressed isothermally from A to B, as the graph illustrates. What is the work involved if the temperature of the gas is 307 K? Be

P) Thermodynamics, PDF, Gases
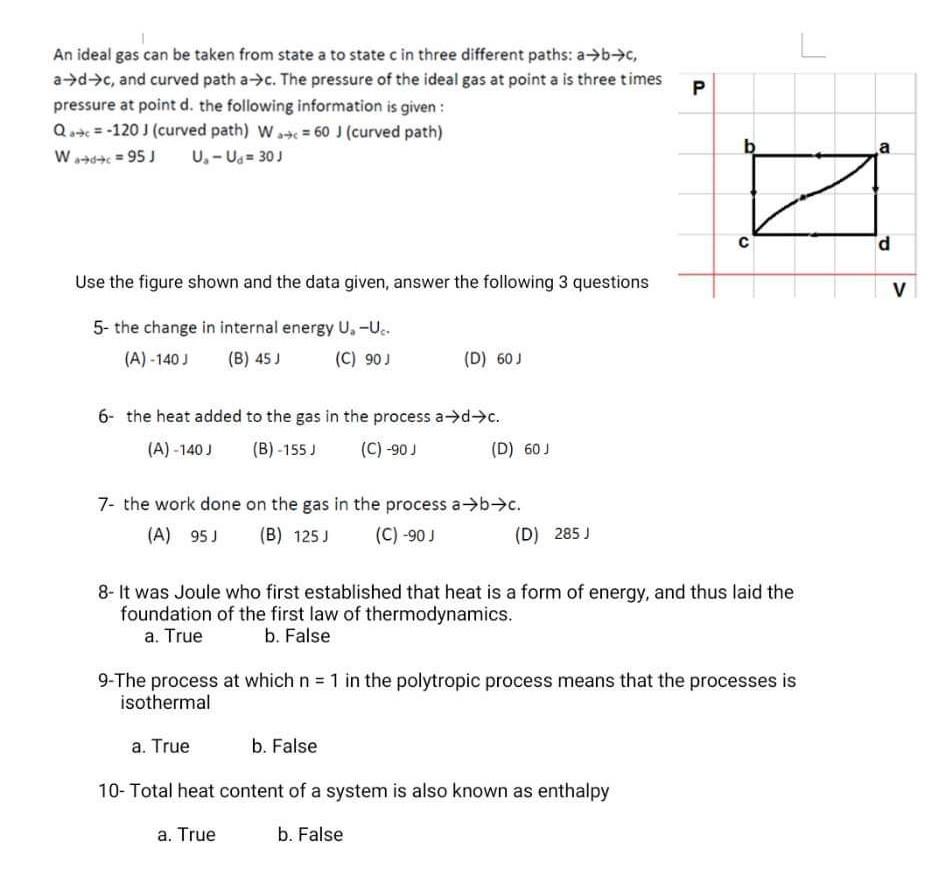
Solved An ideal gas can be taken from state a to state c in
from
per adult (price varies by group size)







