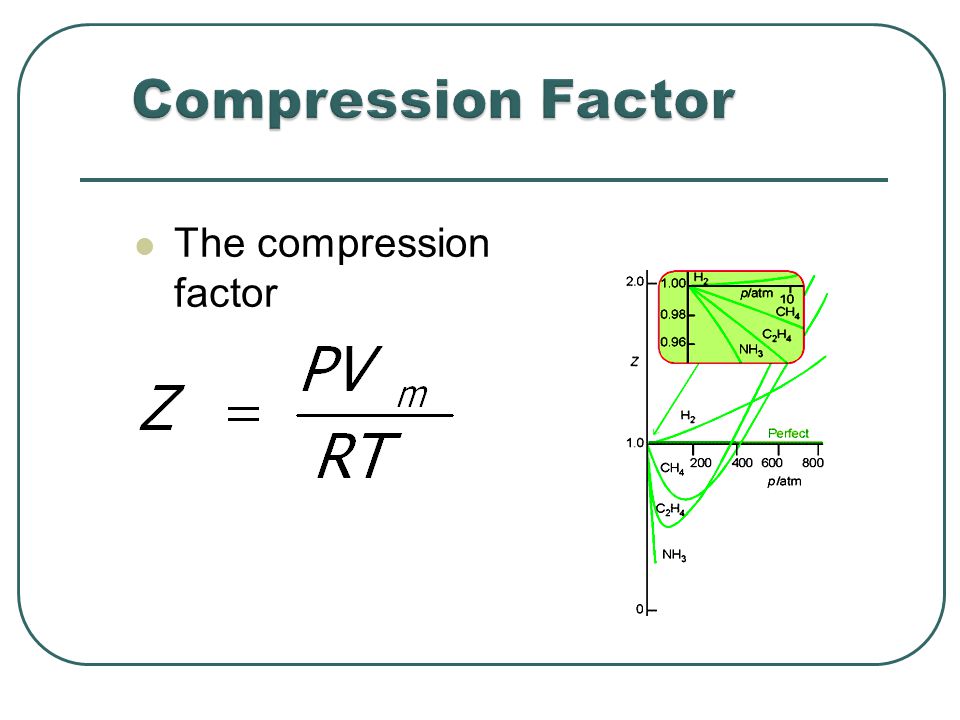
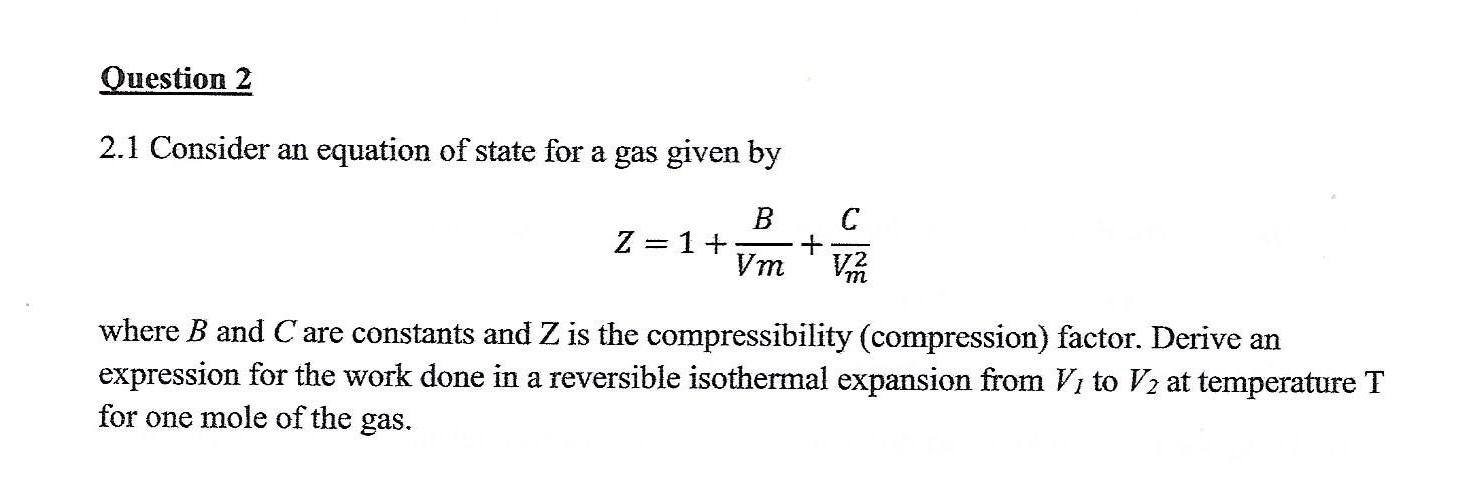the compression factor one mole of a vander waals gas 0 C and 100
By A Mystery Man Writer
Description
Click here:point_up_2:to get an answer to your question :writing_hand:the compression factor for one mole of a vander waals gas at 0 c and
Click here👆to get an answer to your question ✍️ The compression factor one mole of a vander waals gas 0 C and 100 atm pressure is found to be 0-5
Click here👆to get an answer to your question ✍️ The compression factor one mole of a vander waals gas 0 C and 100 atm pressure is found to be 0-5
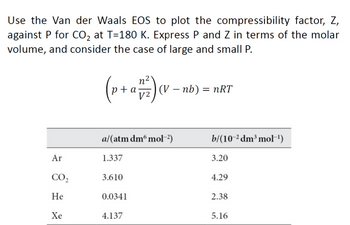
Answered: Use the Van der Waals EOS to plot the…

The compression factor (compressibility factor) for one mole of a Van der..

The compression factor (compressibility factor) for 1 mol of a van der

The compressibility factor 1 mole of vanderwaal gas 0^{o}C, and 100 atm pressure is found to be 0.5, then calculate the vander Waals constant a. Assuming: that the volume of gas molecule

Bengali] The compresibility factor (Z) of one mole of a van der waals

Physical Chemistry The Compression Factor (Z) [w/1 example]

123. The compra compressibility factor one mole of a der Waals gas 0º C and 100 atm pressure found to be 0.5 Assume that the volume of es molecule is negligible. The

The figure displays the plot of compression factor Z versus p a few gases. Which of the following statements is/are correct a van der Waals gas? HA The plot of I is

The figure displays the plot of compression factor Z versus p a few gases. I .-- - --- ------ III II p > Which of the following statements is/are correct a van
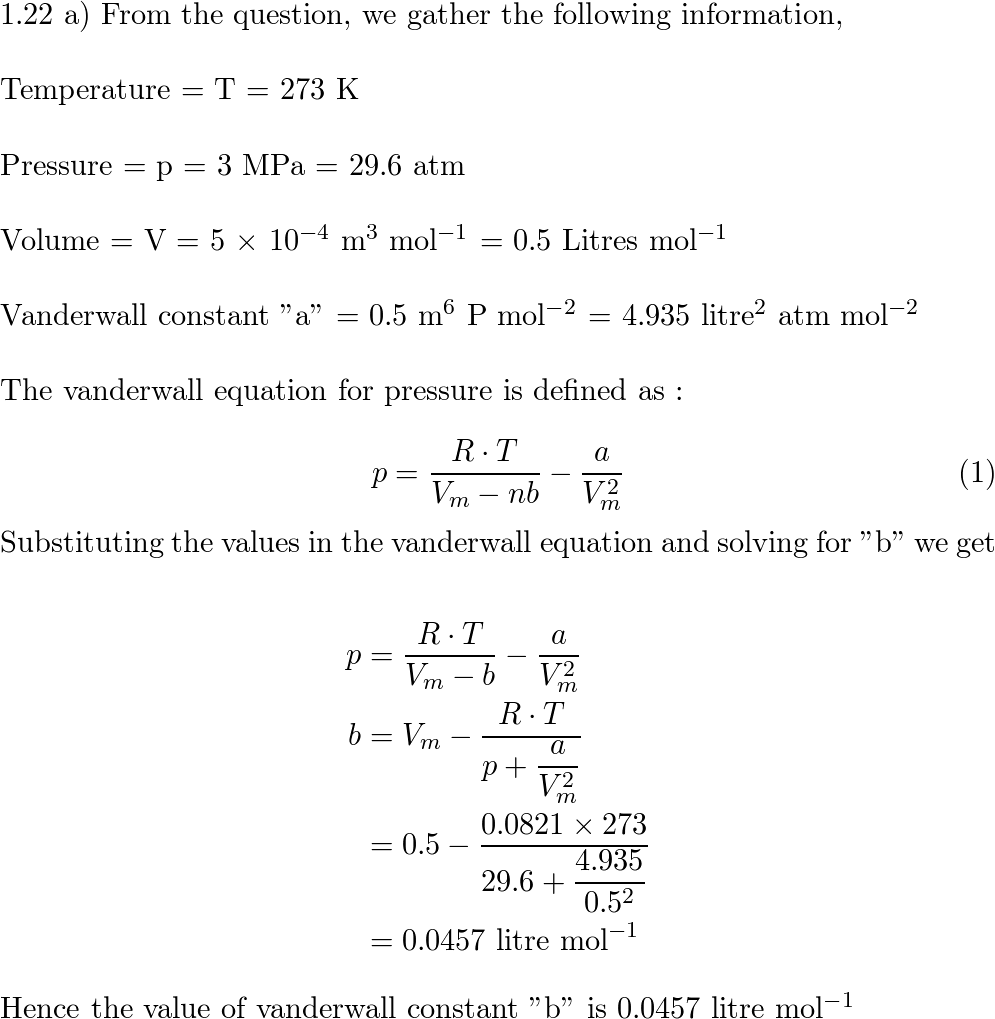
a) A certain gas obeys the van der Waals equation with $a =

18. The compressibility factor one mole of a vanderwaal's gas 0°C and 100 atm pressure is found to be 0.5. Assume that the volume of gas molecule is negligible calculate the vanderwaals

Answered: Compression factor of a gas with van…
from
per adult (price varies by group size)