2.t 300 K, 36 g of glucose present per litre in itssolution has an
By A Mystery Man Writer
Description
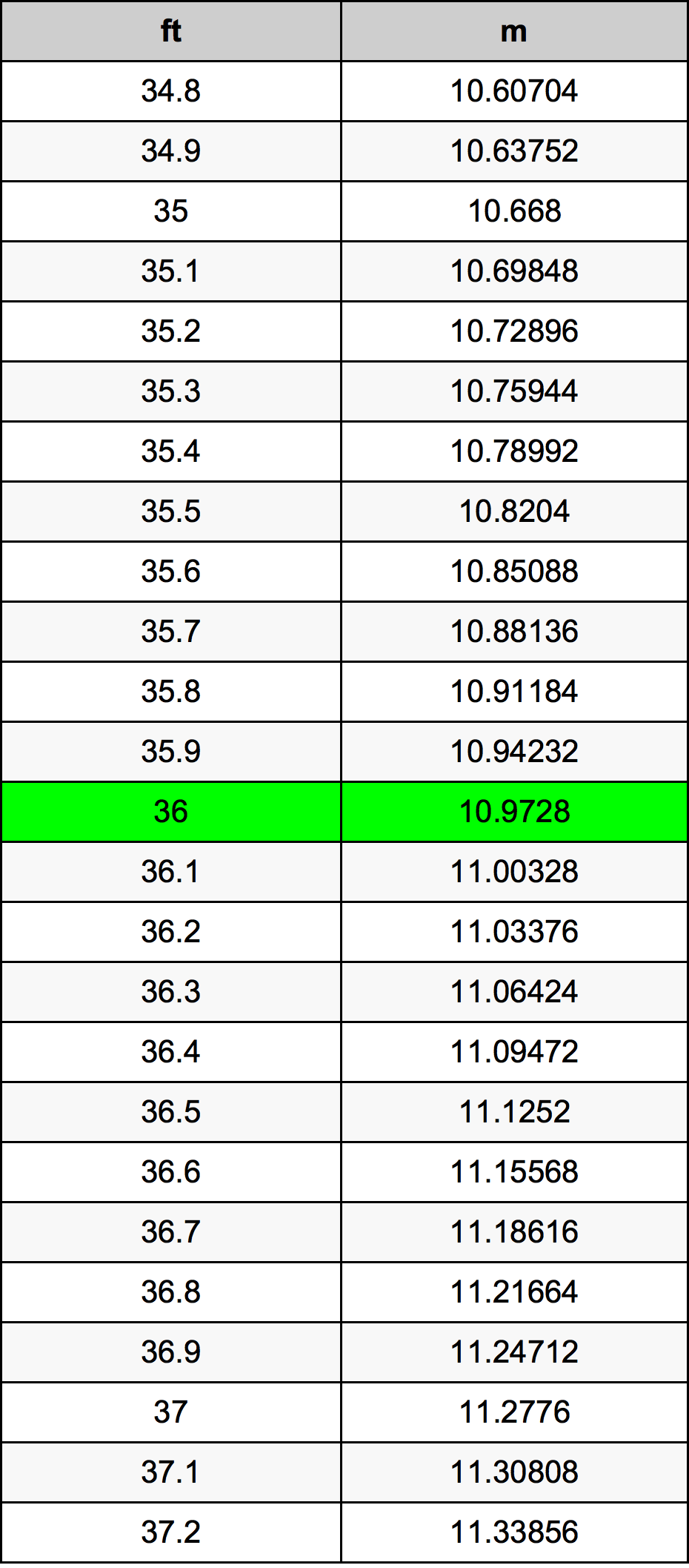
2.t 300 K, 36 g of glucose present per litre in itssolution has an osmotic pressure of 4.98 bar. If theosmotic pressure of solution is 1.52 bar at thesame temperature, what would be itsconcentration?(1) 11 gl 1(3) 36 gl 1(2) 22 gL 1(4) 42 gL 1
2-t 300 K- 36 g of glucose present per litre in itssolution has an osmotic pressure of 4-98 bar- If theosmotic pressure of solution is 1-52 bar at thesame temperature- what would be itsconcentration-1- 11 gl-1-3- 36 gl-1-2- 22 gL-1-4- 42 gL-1
2-t 300 K- 36 g of glucose present per litre in itssolution has an osmotic pressure of 4-98 bar- If theosmotic pressure of solution is 1-52 bar at thesame temperature- what would be itsconcentration-1- 11 gl-1-3- 36 gl-1-2- 22 gL-1-4- 42 gL-1
What is the molarity of a solution containing 16 grams of glucose for a 300 ml solution? - Quora

At 300K, 36g of glucose present in a litre of its solution has an osmotic pressure of 4.98bar.

At 300K, 26g of glucose present in a litre of its solution has an osmotic pressure of 4.98 bar.
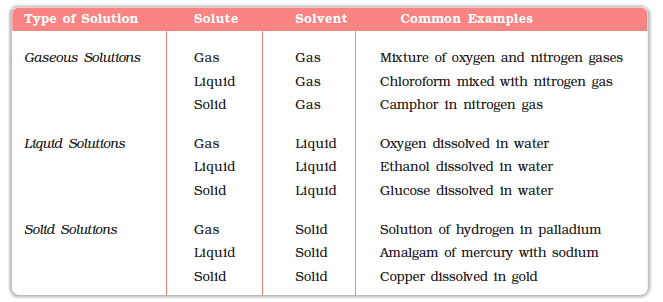
NCERT Ebook for Solutions - Solutions - Chapter 2 - NCERT Chemistry - XII

NCERT Solutions For Class 12 Chemistry Chapter 2 Solutions

Board Que 2023, PDF, Coordination Complex
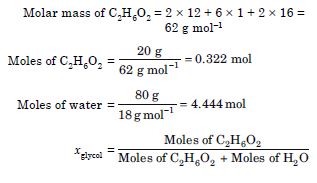
NCERT Solutions for Class 12 Chemistry Solutions

At 300 K, 36 g of glucose present per litre in its solution has an osm

2.22At300 K,36 g of glucose present in a litre of its solution has an osm..

Chemistry Class 12 NCERT Solutions Chapter 2 Solutions – Class 12 Chemistry
from
per adult (price varies by group size)