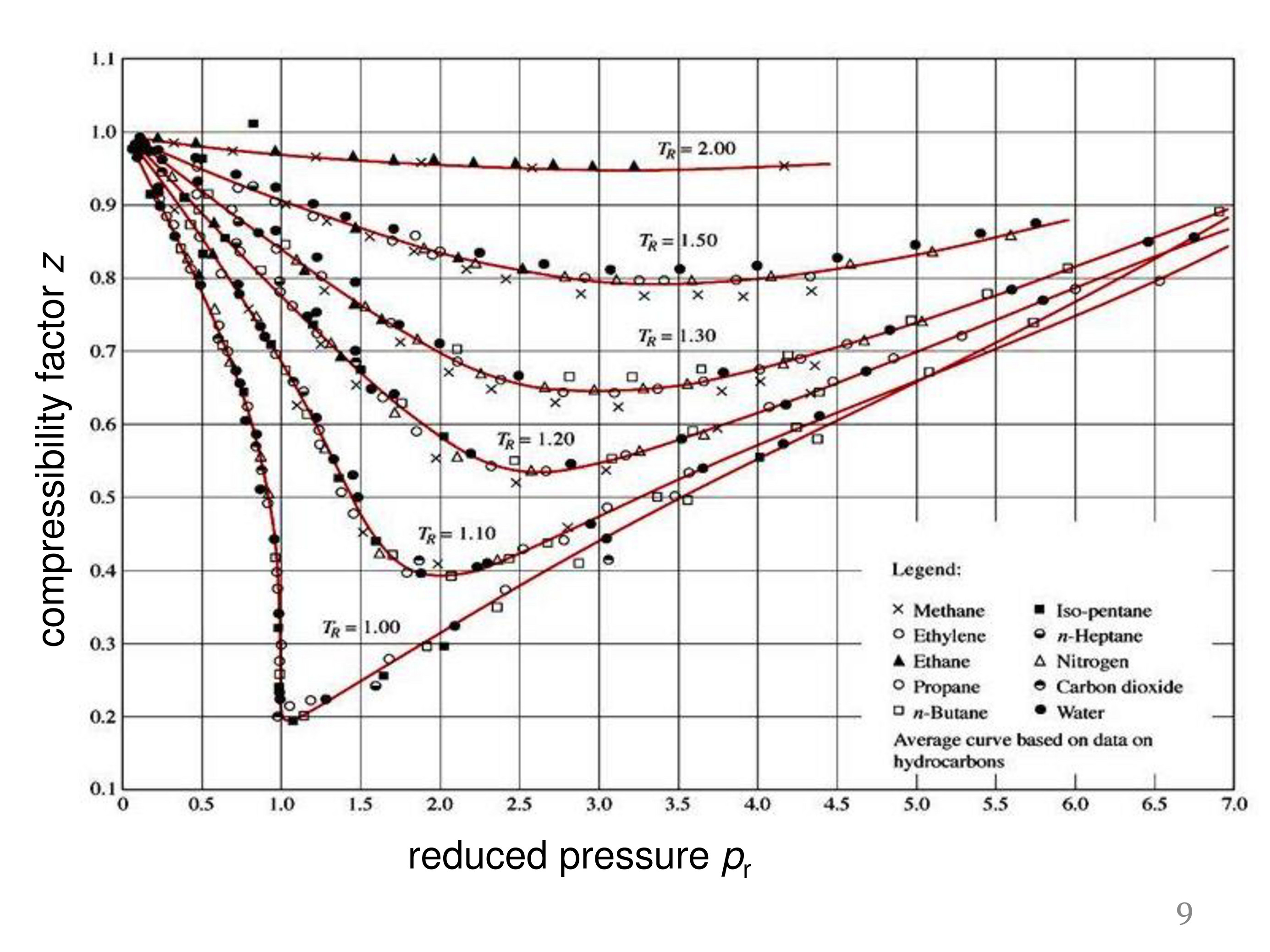
Compressibility factor (Z) is plotted against pressure at different te
By A Mystery Man Writer
Description
Compressibility factor (Z) is plotted against pressure at different temperature for same gas Which of the following is the correct order of temperature s
Is z (compressibility factor) vs P (pressure) graph drawn by changing volume? If it is why it isn't drawn by changing mole - Quora

The compressibility factor (Z) vs P one mole of a real gas is shown in following diagram. The graph is plotted constant temperature 300 K. If the slope of the graph very

gas laws - Graph of compressibility factor vs pressure when real gas is assigned Z=1 - Chemistry Stack Exchange

e Compressibility factor (Z) for hydrogen WRT pressure and temperature

Compressibility factor Z is plotted against pressure P for four different gases A , B , C & D. The correct order of critical temperature of the gases shown in the below

The graph of compressibility factor (Z) v/s P 1 mol of a real gas is shown in following diagram. The graph is plotted 273 K temperature. If slope of graph very high

Energies, Free Full-Text

Correct order of compressibility is

Malayalam] If (A) is correct, but (R) is incorrect.
At Critical Temperature,pressure and volume . The compressibility Factor (Z) Is

Compressibility factor Z = PV / nRT is plotted against pressure as shown below:What is the correct order for the liquefiability of the gases shown in the above graph? A. CO 2
from
per adult (price varies by group size)