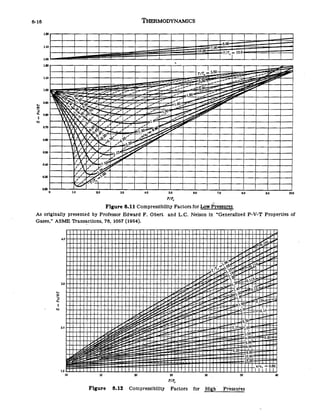
The given graph represent the variations of Z Compressibility factor Z PV nRT versus p for three real gases A B and C Identify the only incorrect statement
By A Mystery Man Writer
Description

4.2: Real Gases (Deviations From Ideal Behavior) - Chemistry LibreTexts
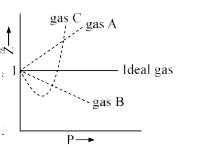
Gas C is a real gas and we can find 'a' and 'b' if intersection data i

Van der Waals Equation - Derivation, Relation Between Ideal Gas Law, Application
Where is the deviation of a gas from ideal behaviour maximum at? - Quora

Compressibility Factor Z Important Concepts and Tips for JEE Main

5. Refer to the figure given : (2006 - 5M; andndash;1) Which of the following statements is wrong?a)For gas A, a = 0 and Z will linearly depend on pressureb)For gas B

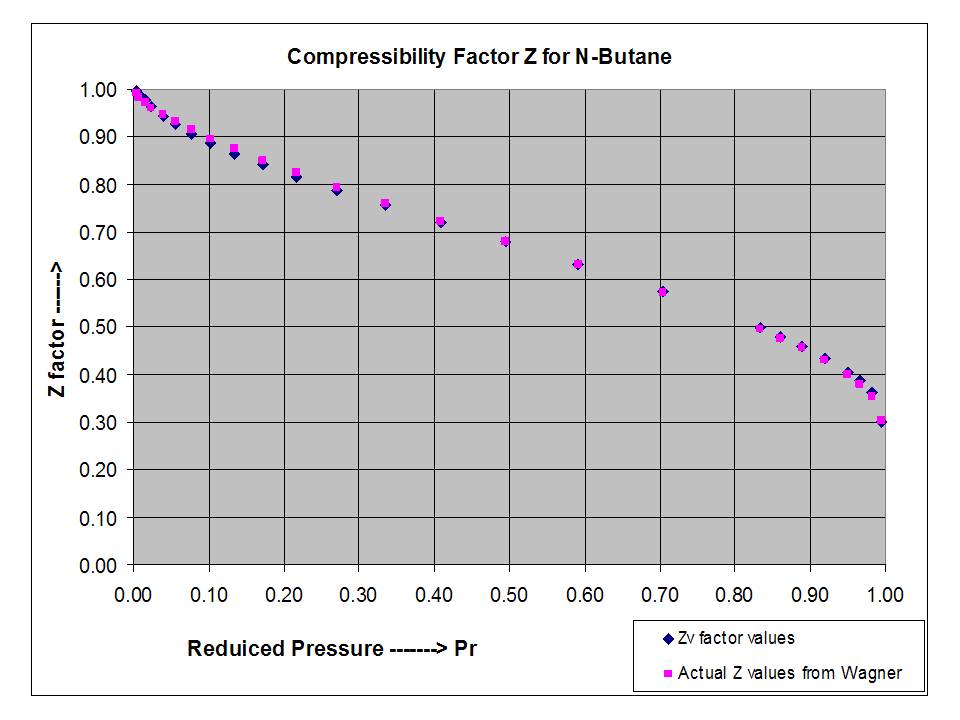
Compressibility Factor of Gas, Overview, Equation & Chart - Lesson

The given graph represents the variations of compressibility factorZ= pv//nRT vs P for three rea

the given graph represents the variation of Z (compressibility factor =dfrac{PV}{nRT}) versus P, three real gases A, B and C. Identify the only correct statement.For the gas A, a = 0 and

Compressibility Factor - an overview

physical chemistry - Why do some gases have lower value of Z for a particular pressure? - Chemistry Stack Exchange

In the figure representing variation of the compressibility factor Z of a real gas with pressure
from
per adult (price varies by group size)