Which of the following statements is/are correct? (a) all real gases are less compressible
By A Mystery Man Writer
Description
Which of the following statements is/are correct? (a) all real gases are less compressible than ideal gas at high pressures? (6) hydrogen and helium are more co
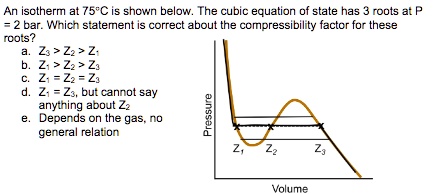
SOLVED: Thermodynamic question: Please answer the multiple choice with an explanation. An isotherm at 75°C is shown below. The cubic equation of state has 3 roots at P = 2 bar. Which
Modeling ideal gas flow using Bernoulli's equation

Select the correct statements among the following: Boyle's temperature ==xInversion temperature At Boyle's temperature, a real gas behaves like ideal gas A real gas behaves like ideal very low pressure and very
Energies, Free Full-Text

What is compressible liquid? : r/thermodynamics

The following graph is plotted between compressibility factor Z versus pressure of a gas at different temperatures.Which of the following statements is /are correct?

Numerical study of real gas effects during bubble collapse using a disequilibrium multiphase model - ScienceDirect

For A Real Gas At 25∘C Temperature And High Pressure (99, 59% OFF

Comparative study on the H2S gas-sensing properties of graphene

Determine which of the following statements are true at very high pres

Consider the equation, Z=dfrac{PV}{nRT}. Which of the following statements is correct?When Z>1, real gases are easier to compress than the ideal gasWhen Z>1, real gases are difficult to compressWhen Z=1, real gases

Gen Chem: Compressibility? : r/Mcat
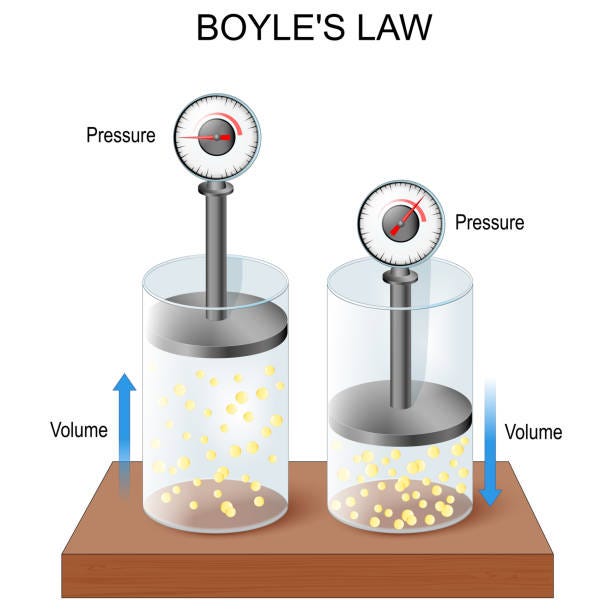
Boyle's law: volume and pressure
Except H(2) and He, the compressibility factor Z(=(PV)/(nRT))lt1 for a

PPT - 6 Gases PowerPoint Presentation, free download - ID:352441
from
per adult (price varies by group size)